Yeast Viability
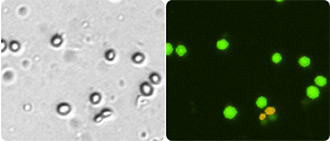
Determining yeast viability and concentration during biofuel and brewing production is essential for process optimization and monitoring of alcohol production. Many of these complex yeast samples increase the difficulty for automated counting using bright field method as well as manual counting due to the debris particles in the sample. By using a combination of fluorescent dyes acridine orange (AO) and propidium iodide (PI) Cellometer instruments can accurately determine the concentration and viability of the complex samples.
Acridine orange (AO) and propidium iodide (PI) are nuclear staining (nucleic acid binding) dyes. AO is permeable to both live and dead cells and stains all nucleated cells to generate green fluorescence. PI enters dead cells with compromised membranes and stains all dead nucleated cells to generate red fluorescence. Cells stained with both AO and PI fluoresce red due to quenching, so all live nucleated cells fluoresce green and all dead nucleated cells fluoresce red.
For AO/PI staining and viability determination, 20 µl of live cell sample is mixed with 20 µl the dilution buffer and followed by 40 µl of AO/PI staining solution. 20 µl of stained sample is then added to a Cellometer Counting Chamber and analyzed in <60 seconds using a fluorescent Cellometer instrument. Each instrument automatically reports live / dead cell number, live/dead cell concentration, mean diameter, and percent viability for the sample tested.
ViaStain™ Yeast Kit for Live/Dead Concentration
| Description | Catalog Number | Unit Size | Manual | SDS | |
|---|---|---|---|---|---|
| AO/PI Yeast Viability Kit containing dilution buffer and fluorescent dye mixture for staining of live and dead Yeast cells. | CSK-0102-2mL
CSK-0102-10mL |
2 mL
10 mL |
US:AO/PI EU:AO/PI |
Buy Online | |
| PI (propidium iodide) staining solution for staining of dead nucleated cells. | CS1-0109-5mL | 5 mL | US:PI EU:PI |
Buy Online | |
| Yeast dilution buffer for use with CSK-0102 (yeast kit for live / dead staining) | CS0-0110-500ML | 500 ML | US:Buffer EU:Buffer |
Buy Online |
ViaStain™ CFDA-AM Yeast Vitality Stain
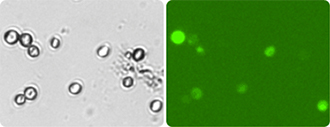
The ViaStain™ CFDA-AM Yeast Vitality Stain enables the user to quantitatively distinguish metabolically active Lager and Ale yeast in pure cultures and in cultures containing debris such as beer slurries using the Cellometer system. The stain contains a solution of a cell-permeant esterase substrate, CFDA-AM (5-Carboxyfluorescein Diacetate, Acetoxymethyl Ester), that fluoresces green when hydrolyzed by enzymatically active Lager and Ale yeast. The percent of yeasts that are actively fermenting during production can be determined and used to optimize the fermentation process during beer brewing, for example.
ViaStain™ CFDA-AM Yeast Vitality Stain
| Description | Catalog Number | Unit Size | Manual | SDS | |
|---|---|---|---|---|---|
| Yeast Vitality Stain enables breweries to detect metabolically active Lager and Ale yeast at different fermentation stages. PBS included. | CSK-0125-200uL | 200 uL | US:CFDA-AM EU:CFDA-AM |
Buy Online |

