Wound Healing
Perform a Wound Healing Migration Assay by Monitoring and Quantifying Cell Growth in a Defined Area
- Measure the confluence and cell count of cells migrated into the exclusion (scratch) zone
- Acquire multiple images of the same plate to produce a kinetic reading of wound healing
- Perform wound healing assay using bright field and/or fluorescent imaging
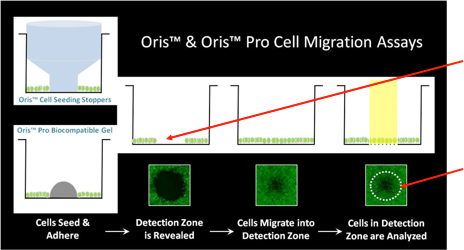
Celigo Wound Healing Application using the 96-Well Platypus Oris™ Plates
Removing the well stopper leaves an exclusion zone into where the cells migrate.
The Celigo detects cell migrating into the exclusion zone either in bright field or fluorescence.
Protocol
- Seeded cells overnight
- Removed the well stopper from the Oris™ plate (alternatively, perform a manual scratch)
- Wells were imaged using Celigo green channel (various exposure time for dim or bright GFP cells)
- Plate was acquired at: 0h, 19h, 24h, 43h and 71h post stopper removal
- Analysis
- Wound Healing application was setup with a 36% well mask reduction to define the analysis area
- Acquisition and analysis parameters were saved to re-use for all time points
Plate Map: Cell Type and Conditions
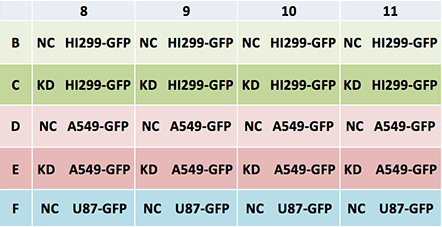
Legend: NC – Negative Control, KD – Knockdown
Measuring the Migration of GFP-Labeled Cells using Platypus Oris™ 96-Well Plates
Whole-well images are acquired and the analysis area is defined by the Celigo software.
The Celigo Wound Healing application reports the wound healing confluence and cell count for each well over time within the defined analysis area.
Whole well fluorescent image
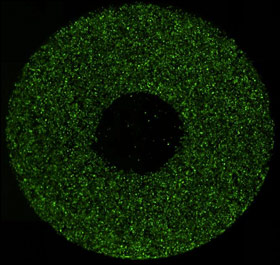
Plate cells. Remove stopper and Image whole well in fluorescence
Definition of analysis area
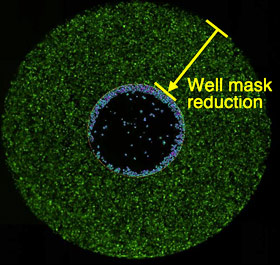
Analysis area is defined by reducing well mask size (36% of the total well area).
Analysis area (Zoomed)
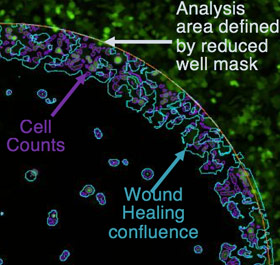
Celigo measures and reports the confluence and cell count within the analysis area.
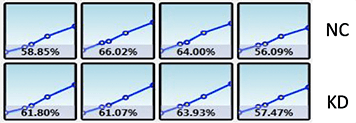
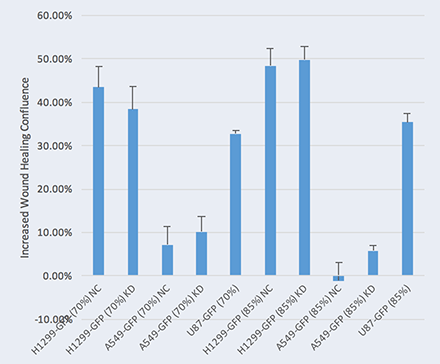
Wound Healing Quantification of GFP-Labeled H1299 Cells
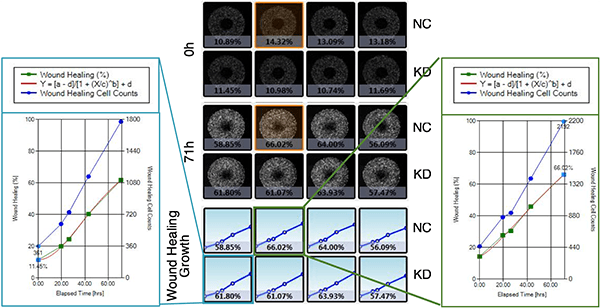
Celigo generated growth curves at the well and plate level for H1299-GFP cells
Increased Wound Healing % = Wound Healing % (71h) – Wound Healing % (0h)
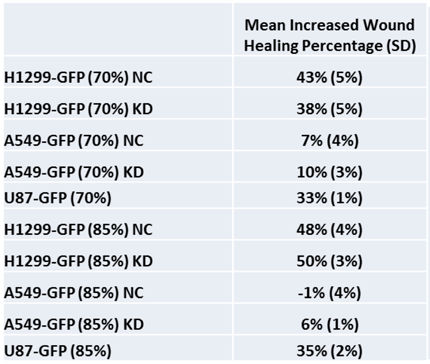
Two seeding densities were tested: 70% and 85%. These seeding densities refer to the confluence of the original plate that was used for seeding onto the Oris plate.
Data indicates that similar wound healing trends were seen for both 70 and 85 percent confluent samples.
In the table below, A549 cells appear to have migrated the least into the wound compared to other cell types in the study.
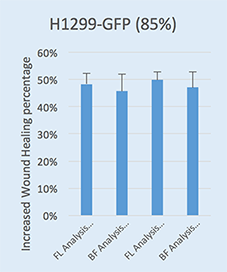
Comparison of Wound Healing Confluence Between Bright Field and Fluorescence Analysis
Bright Field Analysis
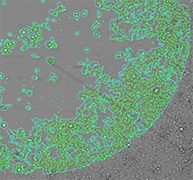
Bright Field H1299-GFP (85%)
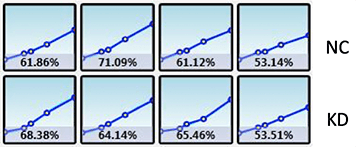
Celigo generated growth curves at the well and plate level for H1299-GFP cells
Fluorescent Analysis
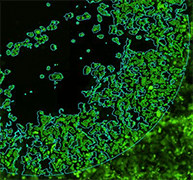
Fluorescent H1299-GFP (85%)