Experimental Procedure
- Combine 20µl of PBMC sample and 20µl of AO/PI dye mixture and mix well by pipetting up and down
- Load 20µl of sample into the disposable counting chamber
- Allow cells to settle in chamber for less than 1 minute
- Insert chamber into Cellometer instrument
- Select assay from menu
- Enter sample ID manually or scan in with bar code reader
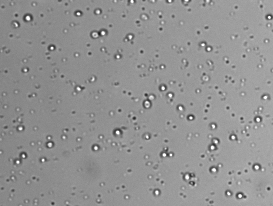
- Preview bright-field cell image and adjust focus (if necessary)
- Click “Count” to begin analysis
- Review images and counting results on-screen
- Images, cell count, concentration, % viability, and cell diameter data are saved to a secure network location
Accurate Cell Count and Viability Results
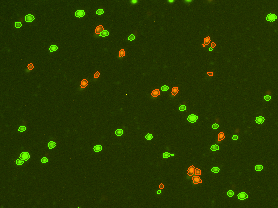
Each PBMC sample is incubated with an acridine orange / propidium iodide (AO/PI) dye mixture. The AO dye stains DNA in the cell nucleus of both live and dead cells. Mature mammalian red bloods cells don’t contain nuclei, so only peripheral blood mononuclear cells are stained, for a more accurate total PBMC count. Propidium iodide DNA-binding dye is used to determine cell viability. Healthy cells are impermeable to the PI dye. Only dead (non-viable) cells with compromised membranes are stained. Cells stained with both AO and PI fluoresce red due to Förster resonance energy transfer (FRET).
- Live nucleated cells are counted in the green channel
- Dead nucleated cells are counted in the red channel
- There is no interference from platelets or red blood cells
In less than 60 seconds, the Auto 2000 reports live cell count and concentration, dead cell count and concentration, mean cell diameter, and % viability for the sample. Bright field and fluorescent cell images can be viewed, saved, and exported for presentation or publication. Download the full Nexcelom PBMC Application Note for more information.


Leave A Comment