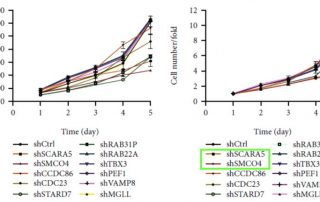
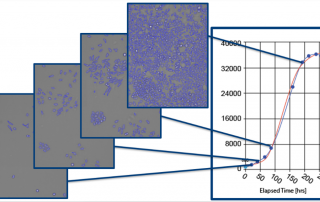
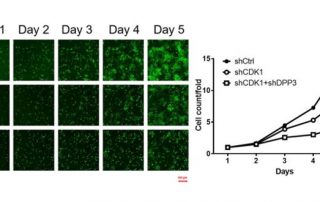
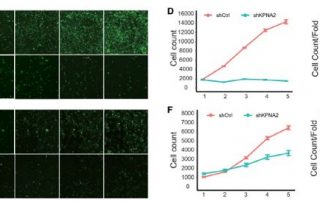
SCARA5 Promotes Proliferation and Migration of Esophageal Squamous Cell Carcinoma
Exploration of candidate genes responsible for esophageal squamous cell carcinoma (ESCC) pathogenesis may provide insight into the underlying signaling pathways to uncover novel therapeutic targets.